The Fundamental Architecture of Nuclear Stability

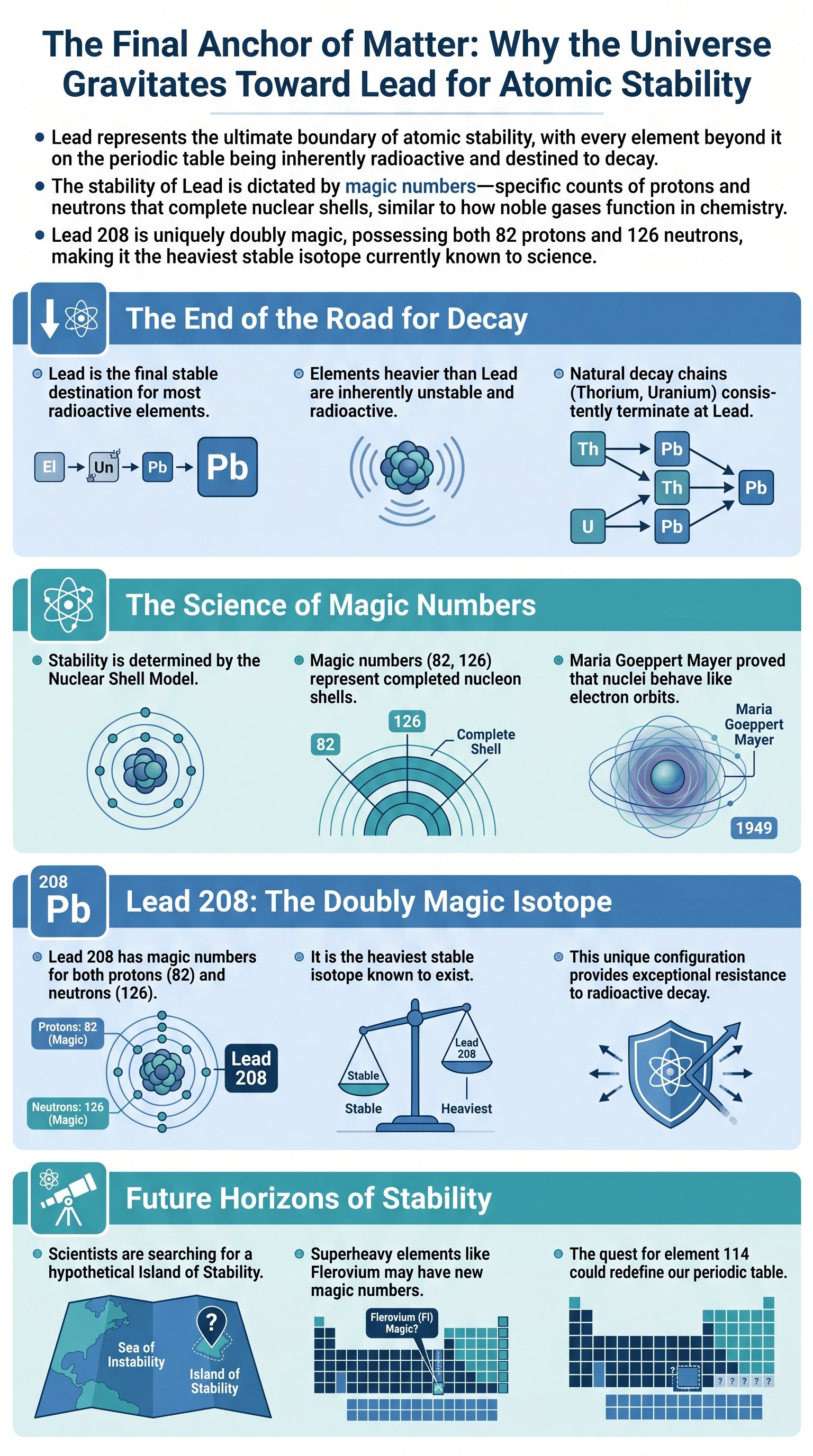
In the vast landscape of the periodic table, Lead occupies a position of profound significance. To understand why nature seems to favor this heavy metal, we must first look at the core of the atom: the nucleus. Composed of protons and neutrons—collectively known as nucleons—the nucleus is the engine room of an element's identity. While the number of protons defines the element itself, the balance between these particles determines whether an atom will endure for eternity or eventually break apart.
Every atom with 82 protons is categorized as Lead. However, the number of neutrons can vary, creating different isotopes. Some isotopes are stable, meaning they can exist indefinitely without changing. Others are radioactive, constantly shedding particles in a desperate search for a more balanced configuration. This process, known as radioactive decay, is the mechanism by which the universe filters out instability.
Lead 208 is the pinnacle of this process. It contains 82 protons and 126 neutrons. In the realm of nuclear physics, Lead is not just another element; it is often referred to as being magic. This term describes the phenomenon where specific numbers of nucleons lead to significantly higher stability than their neighbors on the periodic table.
Key insight: The stability of an element is not random; it is the result of nucleons filling specific energy levels or shells within the nucleus, reaching a state of equilibrium that resists external disruption.
Historically, humans have been fascinated by Lead's properties, from the ancient Romans using it as a sweetener to modern medical professionals using it for X-ray shielding. Yet, the true marvel lies in its role as the final destination for the most volatile elements in existence.
| Element Type | Proton Count | Stability Status |
|---|---|---|
| Lead (Stable Isotopes) | 82 | Ultimate End Point |
| Polonium | 84 | Radioactive |
| Uranium | 92 | Radioactive |
The Mechanics of Alpha and Beta Decay Chains

Radioactivity is essentially the language of atomic transition. When a nucleus is too heavy or has an unfavorable ratio of protons to neutrons, it undergoes decay. The two primary methods discussed by Reid Reimers are Alpha decay and Beta decay. Alpha decay involves the ejection of a helium-4 nucleus—two protons and two neutrons—which significantly reduces the size of the original atom.
Beta decay is more subtle. It involves the transformation of a neutron into a proton (or vice versa), releasing an electron or positron. While Beta decay doesn't change the total number of nucleons, it changes the atomic number, effectively turning the atom into a different element. One decay event is rarely enough to achieve stability; instead, atoms often embark on long, predictable journeys known as decay chains.
There are three primary decay chains found in nature: the Thorium series, the Actinium series, and the Radium (or Uranium) series. Each of these paths follows a rigorous sequence of transformations, shedding mass and energy over millions or billions of years. Remarkably, almost all of these paths terminate at a stable isotope of Lead.
- 1The Thorium series ends at Lead 208.
- 2The Actinium series ends at Lead 207.
- 3The Radium series ends at Lead 206.
Note: A fourth chain, the Neptunium series, is largely extinct in nature because its isotopes have short half-lives, eventually settling at Thallium 205 instead of Lead.
Thorium 232, for instance, has a half-life of 14 billion years—roughly the age of the universe. Yet, even this incredibly enduring isotope will eventually transition through various elements like Radium and Actinium before finding its final, peaceful state as Lead 208. This consistency demonstrates a universal preference for the Lead configuration.
The Nuclear Shell Model and Magic Numbers
The reason Lead acts as such a powerful attractor for decaying matter was discovered in the 1940s by chemist Maria Goeppert Mayer. While mapping the abundance of isotopes, she noticed that atoms with specific numbers of protons or neutrons were disproportionately stable. These numbers—2, 8, 20, 28, 50, 82, and 126—became known as magic numbers.