The Fundamental Constraint of Light and the Quantum Solution

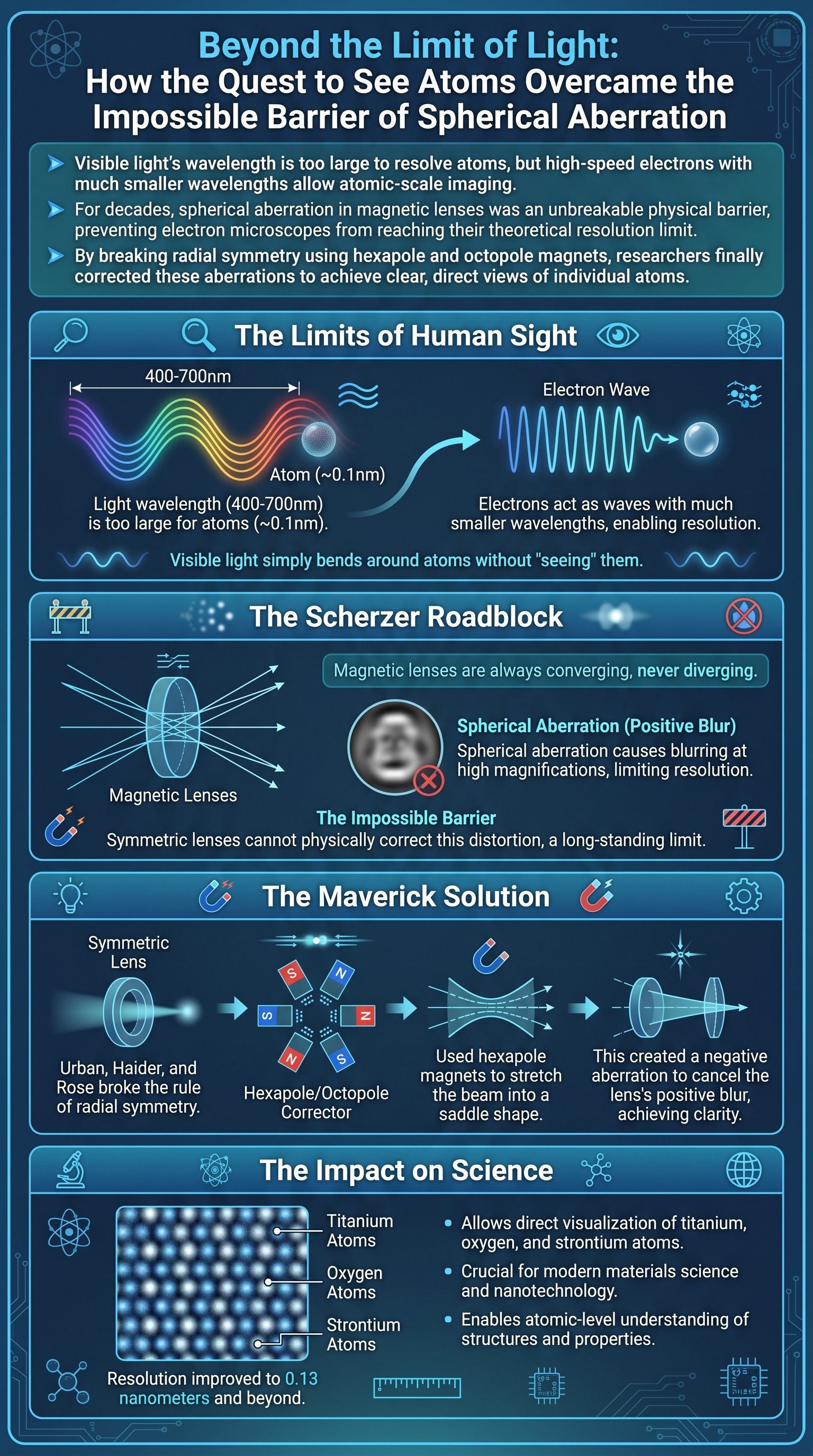
To understand why we cannot see atoms with standard equipment, we must first look at the physics of light. Visible light has wavelengths between 380 and 750 nanometers, while an individual atom is roughly 0.1 nanometers. Because the wavelength of light is over 3,000 times larger than an atom, light waves simply diffract or bend around the atom rather than reflecting off it. To visualize something so small, we require a probe with a significantly smaller wavelength. This challenge remained a fundamental wall for science until the advent of quantum mechanics.
In 1924, French physicist Louis de Broglie proposed a revolutionary idea: matter, like light, has wavelike properties. He calculated that the wavelength of a particle is inversely proportional to its momentum. By accelerating electrons to 80% or 90% the speed of light, scientists can generate a wavelength of approximately 2 to 3 picometers. This is over 100,000 times smaller than visible light, providing the theoretical capability to resolve individual atoms. This discovery paved the way for the development of the electron microscope, shifting the focus from optical lenses to electromagnetic fields.
| Feature | Optical Microscope | Electron Microscope |
|---|---|---|
| Source | Visible Light | High-speed Electrons |
| Wavelength | 380 - 750 nm | ~0.002 nm |
| Lens Type | Glass Lenses | Electromagnetic Coils |
| Max Resolution | ~200 nm | < 0.1 nm |
Key insight: The resolution of any imaging system is fundamentally limited by the wavelength of the probe being used; to see smaller, you must go faster.
The Invention of the Electron Microscope and its Fatal Flaw

Since glass lenses cannot bend electron beams, researchers had to find a new way to focus them. German physicist Hans Busch suggested that an electromagnetic lens could work, but it was Ernst Ruska who built the first prototype in 1931. By coiling wire around an iron core and leaving a small gap, Ruska created a donut-shaped magnetic field. As electrons passed through this field, the Lorentz force caused them to spiral and converge toward a central point. This was the birth of the Transmission Electron Microscope (TEM), which creates an image by passing electrons through a sample.
However, progress soon hit a massive roadblock known as the Scherzer limit. In 1936, Otto Scherzer published a paper proving that all radially symmetric magnetic lenses suffer from unavoidable spherical aberration. This means that electrons passing through the edges of the lens are deflected more strongly than those in the center, causing the focus to blur. In optical cameras, this is fixed by using a diverging lens to cancel the convergence, but Scherzer proved that a diverging magnetic lens is physically impossible due to Maxwell's equations.
- The magnetic field is always stronger at the edges of the lens.
- Electrons are over-deflected, focusing at different points along the axis.
- This creates a 'blur' that gets worse as magnification increases.
- Every radially symmetric magnetic lens is doomed to converge the beam.
Caution: For over 50 years, the scientific community accepted the Scherzer limit as an absolute law of physics that would forever prevent us from seeing atoms clearly.
The Scanning Revolution and the Abandoned Search for Clarity
During the 1960s and 70s, Albert Crewe introduced a new approach to overcome the limitations of the standard TEM. Instead of a broad beam, he used a sharpened tungsten tip to create a narrow, bright beam of electrons that scanned across a sample bit by bit. This became the Scanning Transmission Electron Microscope (STEM). While this method produced the first widely accepted images of single atoms in 1970, the images remained fuzzy and distorted because the underlying problem of spherical aberration still existed.