The Mystery of the Missing Colors: Joseph von Fraunhofer’s Legacy

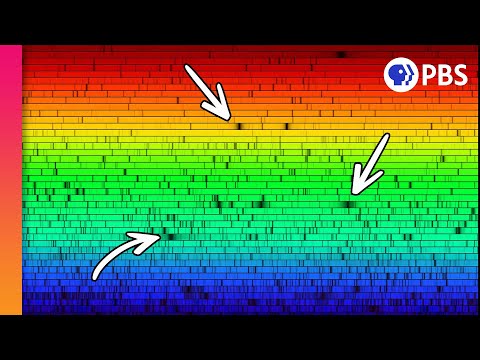
In the early 1800s, the German physicist Joseph von Fraunhofer made a discovery that would change the course of modern astronomy. While observing sunlight passed through a high-quality prism, he noticed something highly unusual: the rainbow was not a continuous band of color. Instead, it was punctuated by hundreds of thin, dark lines where specific wavelengths appeared to be missing. Fraunhofer meticulously cataloged over 600 of these gaps, though he could not explain their origin at the time.
These dark bands looked remarkably like a modern barcode, and as it turned out, that is exactly what they were. For decades, the scientific community puzzled over these missing pieces, which appeared consistently in the same locations within the solar spectrum. This discovery moved the study of light beyond mere aesthetics and into the realm of quantitative analysis, providing the first hint that light carried encoded data about its source.
Key insight: The gaps in the rainbow are not errors; they are data points that allow us to identify the chemical 'fingerprints' of distant celestial bodies without ever leaving Earth.
Fraunhofer's work established the foundation for the field of spectroscopy. By mapping the exact positions of these lines, he created a reference system that later scientists would use to decode the very nature of matter. His precision in manufacturing optical glass allowed him to see what others had missed, proving that even the most mundane phenomena, like sunlight, contain deep layers of hidden information.
Elemental Fingerprints: The Bunsen and Kirchhoff Breakthrough

The breakthrough required to decode Fraunhofer’s mystery came nearly 50 years later from two researchers at the University of Heidelberg: Gustav Kirchhoff and Robert Bunsen. They were obsessed with the way different chemical elements emitted distinct colors when placed in a flame. To study this more precisely, they developed the spectroscope, an instrument that channeled light through a narrow slit and a prism to measure individual wavelengths.
They discovered that when elements like sodium or lithium were heated, they didn't produce a full rainbow, but rather specific bright lines at fixed intervals. Crucially, they realized that the bright yellow lines produced by burning sodium were in the exact same positions as two prominent dark lines in Fraunhofer’s solar spectrum. This led to the realization that elements have a dual nature: they emit specific light when heated and absorb that same light when placed in front of a continuous source.
| Process Type | Visual Appearance | Cause |
|---|---|---|
| Emission | Bright colored lines on black | Electrons releasing energy as they drop to lower states. |
| Absorption | Dark lines on a rainbow | Electrons stealing specific photons to jump to higher states. |
This duality meant that the dark lines in the sun's spectrum were caused by elements in the cooler solar atmosphere absorbing specific frequencies from the hotter core. By comparing the 'barcodes' of known elements on Earth with the lines found in sunlight, scientists could finally determine what the sun was made of—hydrogen, helium, sodium, and more—from 150 million kilometers away.
Check: No two elements produce the same spectral pattern, making spectroscopy the most reliable method for chemical identification in the universe.
The Quantum Engine: How Electrons Manipulate Photons
To understand why elements are so picky about the light they interact with, we must look at the atomic structure. Every atom consists of a nucleus surrounded by electrons residing in specific energy levels. These electrons cannot exist between levels; they must occupy one or the other. When an electron receives exactly the right amount of energy—often in the form of a photon—it jumps from its ground state to a higher, unstable energy state.