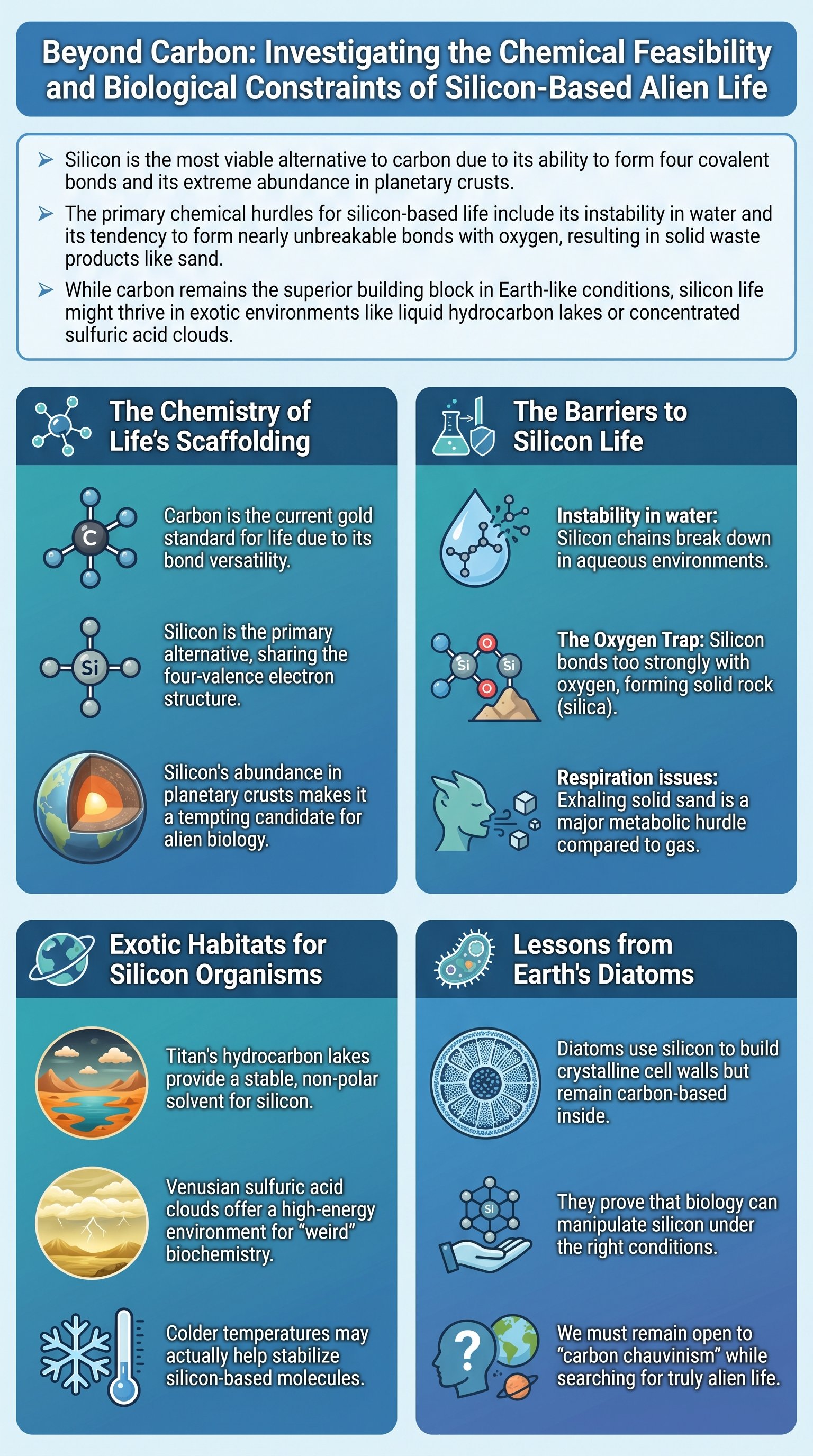
The Chemical Monopoly of Carbon and the Silicon Alternative

All biochemistry as we currently understand it is built upon the carbon atom. Carbon serves as the fundamental scaffolding for DNA, RNA, amino acids, and proteins, creating the complex molecular machinery that defines life. In the realm of astrobiology and science fiction, the most popular alternative is silicon. This is not mere fantasy; silicon resides directly below carbon on the periodic table, meaning it shares the critical property of having four valence electrons. This allows silicon to form four covalent bonds, theoretically enabling it to create long chains and complex rings similar to those found in organic chemistry.
Despite its chemical similarity, silicon is vastly underutilized by Earth's biology. While silicon makes up approximately 28% of the Earth's crust, carbon accounts for a mere 0.02%. This disparity raises a profound scientific question: if silicon is so abundant and chemically versatile, why did evolution choose carbon? The answer lies in the delicate balance of reactivity and stability. For life to function, its chemical bonds must be strong enough to maintain structure but weak enough to be rearranged during metabolic processes. Life is essentially an ongoing chemical reaction that must avoid reaching a state of permanent equilibrium.
Key insight: Carbon's utility comes from its ability to form strong, yet reversible, bonds with itself and other elements, maintaining the 'Goldilocks' zone of chemical energy.
| Feature | Carbon | Silicon |
|---|---|---|
| Valence Electrons | 4 | 4 |
| Crust Abundance | 0.02% | 28% |
| Primary Bond Type | Covalent | Covalent |
| Oxide State | Gas (CO2) | Solid (SiO2) |
The Thermodynamic Balance: Reactivity vs. Stability

To understand why carbon is favored, we must look at how nature seeks the lowest energy state. High-energy molecules like nitroglycerin are unstable because they release energy explosively to reach a more stable configuration. Life requires molecules that can transition between these states without being unidirectional. If a reaction only goes in one direction toward a super-stable molecule, the chemical process stops, and in biological terms, the organism dies. Carbon-based molecules provide this modularity, allowing for the constant recycling of materials through metabolic pathways.
When we examine the periodic table, we can rule out most elements for the basis of life. Ionic and metallic bonds lack the variety and modularity needed for complex structures. Covalent bonds are the only viable option. While silicon can form these bonds, its larger atomic radius makes its bonds with hydrogen and other elements weaker than carbon's. This leads to structural fragility in many complex silicon-based molecules. Furthermore, Rows 4 and below in the periodic table are generally unsuitable because their nuclei cannot hold onto valence electrons with enough strength to form resilient biochemistry.
- 1Carbon forms stable 1D and 2D structures with itself.
- 2Silicon can imitate these structures but often lacks the same degree of stability.
- 3Row 3 elements like Silicon and Phosphorus are the edge of what is structurally viable.
- 4Row 4 elements like Arsenic are toxic precisely because they create weak, unstable analogs of necessary molecules.
Caution: Silicon molecules are often far more reactive with water than their carbon counterparts, making them fundamentally incompatible with Earth's aqueous environment.
The Solvent Crisis: Why Water Rejects Silicon
One of the most significant barriers to silicon-based life is the presence of water. Water is the universal solvent for Earth's life, providing a medium for molecular machinery to interact. However, most complex silicon molecules are unstable in water and tend to break down rapidly. This chemical intolerance means that if silicon life exists, it almost certainly did not evolve in a water-based environment. This leads astrobiologists to consider alternative solvents that could accommodate silicon chemistry.